RU-486 abortion pill approved by Health Canada
Doctor's prescription will be required to purchase drug
The abortion drug known as RU-486 has received the green light from Health Canada.
The regulator said late Wednesday it has approved the use ofMifegymisoto terminate pregnanciesup to a gestational age of 49 days.
Canadian women will need to obtain a prescription from a doctor to purchase the combinationdrug.
- RU-486: What you need to know
- Abortion drug decision pushed back by Health Canada
- Tom Mulcair warns Rona Ambrose against politics in RU-486 application
- Abortion debate may return as Health Canada weighs RU-486 approval
"Health Canada confirms that this decision has been taken and that the company has been informed yesterday," a spokesmansaid in an email.
The product will be distributed by Celopharma Inc. and is expected to be available for sale inwinter 2016, saida statement from the drug's manufacturer,London, U.K.-basedLinepharmaInternational Limited.
"The decision does not rest with me,"Health MinisterRonaAmbrose said Thursday from St. Albert, north of Edmonton. "It's out my hands and the decision is final."
The drug has been available since 1988 in France. The drug was approved for use in the United States in 2000 and is also available inmore than 57 countries,Linepharma said.
While itis often calledmifepristone, mifegymisocontains two drugs,mifepristoneandmisoprostol. The former blocks production of the hormone progesterone, needed to sustain a pregnancy. The latter prompts the uterus to contract and expel the placenta and the fetus.
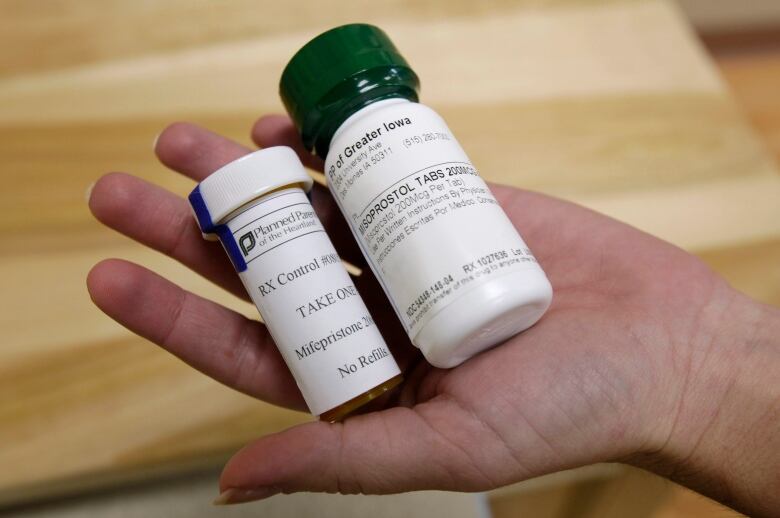
Canadian womenwill take one pill at a doctor's office, go home and take four pills within 12 to 24 hours and then return to the doctor one to two weeks later for a followup visit, a spokeswoman forLinepharma said.
VickiSaporta, president and CEO of the National Abortion Federation and its Canadian offshoot, NAF Canada, said the application has been before Health Canada since December 2012.
Gold standard approach
"No one can claim that they fast-tracked the approval process and didn't very thoroughly and completely review the application," Saporta said.
NAF Canada represents health-care professionals who provide most of the abortion care in this country and worked to introduce thedrug in Canada.
Dr. ErikaFeuerstein, a family physician at Women's College Hospital and Bay Centre for Birth Controlin Toronto, said it's more effective and efficient than the current medical option available in Canada a combination of anticancer injectable drug methotrexatefollowed by misoprostol.
"It works a bit faster, it has a higher success rate,"Feuerstein said.
Some women want to have a surgical abortion and other women prefer a medical abortion, Feuersteinsaid. "It's nice that they can have the option to choose which method serves them the best."
Surgical abortion will continue to be important in situations when the medical abortion fails, Feuersteinadded.
Studies suggest the drug can be used safely as late as 70 daysinto a pregnancy.
Dr. AndrUlmann, lead scientist on RU-486at Linepharma, saidthe company always starts with 49 days when seeking market authorizationto be cautious.
Health Canada did not answer questions from CBC News on details ofthe approval.
Dr. Jennifer Blake, CEO of the Society of Obstetricians and Gynecologistsin Ottawa, said both OBGYNs and family physicians who complete training on the safeuse of this medication will be able to prescribe it.
Asked about the timing of the decision, Blake said the society was notified in thewinter about the status of the decision and it was released on schedule.
Rebecca Cook, a law professor in the International Reproductive and Sexual Health Law Program at the University of Toronto, wrote a 2013 commentary in the Canadian Medical Association Journal that was titled "Medical abortion in Canada: behind the times."
"The approval of the mifepristone/misoprostol product by Health Canada is an important achievement for Canada. Science has prevailed over ideology to ensure that women have access to the benefits of scientific progress,"Cook said in a statement to CBC News.
Mifepristonewithmisoprostolis considered the "gold standard"for medical abortions, Cook and her co-author Dr. Sheila Dunn of Women's College Hospital in Toronto said in their commentary. It's included in the World Health Organizations list of "essential medicines" theminimum medicines needed for basic health-care systems, based on criteria such as safety and cost effectiveness.
University of Toronto ethicist Kerry Bowman expects thatwill increase access to abortion for women who live inrural and remote places.
"The hope is that with time it will enter intofurther out there areas. Maybe even midwives, even nurse clinicians. I don't know. I'm not saying that now, butovertime."
With files from The Canadian Press and CBC's Marisa Dragani












_(720p).jpg)


 OFFICIAL HD MUSIC VIDEO.jpg)
.jpg)