Google venture arm backs universal flu vaccine clinical trial
Vaccitech in U.K. raises $27M to advance flu, other vaccines
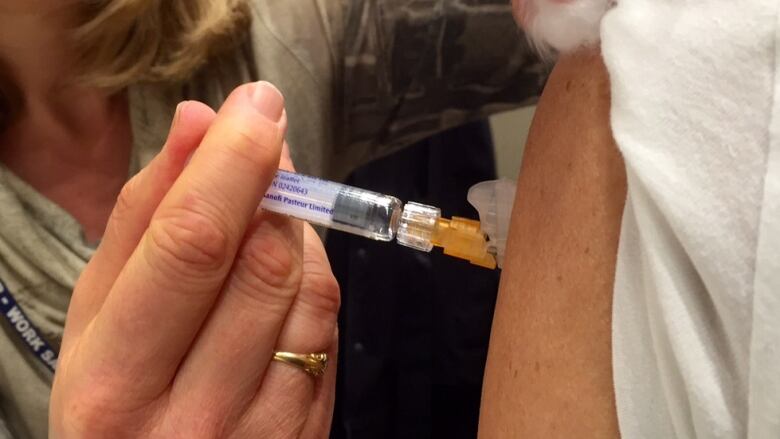
A private British companydeveloping a vaccine that would be the first in the world tofight all types of flu has raised 20 million pounds ($27million US) from investors including GV, the venture capital arm ofGoogle parent Alphabet Inc.
Vaccitech, a spin-out founded by scientists at OxfordUniversity's Jenner Institute, said on Monday the cash wouldhelp fund its vaccine through a two-year clinical trialinvolving more than 2,000 patients and expand other projects.
The group is also running clinical studies on anexperimental shot to prevent Middle East Respiratory Syndrome(MERS) and a therapeutic prostate cancer vaccine for use with animmunotherapy drug. Recent advances have made such vaccine-drugcombinations a hot area of cancer research.
- Flu activity in Canada 'high' and continuing to rise, latest public health numbers say
- Flu season most severe city has seen, says Calgary ER head
A so-called universal flu vaccine that elicits immunityagainst parts of the virus that do not change from year to yearis a holy grail of medicine but so far it has proved elusive.
Current flu vaccines have to be changed each year to matchstrains of virus circulating at the time. The hope is the newone-size-fits-all vaccine will provide better and longer-lastingprotection.
Vaccitech's new vaccine works by using proteins found in thecore of the virus rather than those on its surface. Surfaceproteins stick out like pins from the virus and change all thetime, while those in the core are stable.
Stimulate T-cells
It also stimulates T-cells rather than antibodies anapproach that has yet to convince existing flu vaccinemanufacturers like Sanofi, GlaxoSmithKline andCSL's Seqirus.
- Flu vaccine may have low effectiveness against dominant strain, Canada's top public health doctor says
- Annual flu shot saves the lives of seniors
Still, Vaccitechchief executive Tom Evans is confident thebig players will come around if the current mid-stage clinicaltrial is a success and he will not have a problem in finding apartner to take the product into final-stage Phase III tests.
"If we get positive data that shows we can affect rates ofhospitalisation and illness with influenza then there is noquestion in my mind that a partner would take this on," he toldReuters. "This could be a game-changer in a very competitivemarket."
The Vaccitech trial marks the first time that a universalflu vaccine has progressed beyond Phase I clinical testing.
If all goes well, Vaccitech's shot could potentially beready for launch in 2023, although Evans said 2024 or 2025 mightbe more realistic. That means financial backers need to take along view, especially as its other programs using T-cells tomake vaccines against cancer, MERS, hepatitis B and humanpapillomavirus are also at an early stage.
"We wanted people who were willing to stick with us for awhile and had bigger pockets for doing secondary rounds," Evanssaid.
Vaccitech's latest financing round was also supported bySequoia China and Oxford Sciences Innovation.











_(720p).jpg)


 OFFICIAL HD MUSIC VIDEO.jpg)
.jpg)